Learn how Magnitude Instrument’s noise suppression technology enables transient absorption measurements of halide perovskite photovoltaic materials under device-relevant optical conditions.
Enabling ns-TA Measurements Under Device-Relevant Conditions
Table of Contents
ABSTRACT: Recent innovations in noise suppression technology from Magnitude Instruments have enabled orders of magnitude enhancement in the sensitivity of transient absorption (TA) spectroscopy. Here we show how the sensitivity enabled TA measurements of halide perovskite photovoltaics with photogenerated carrier densities that matched the operating conditions of solar cells and provided new insight about the electronic and transport properties of these remarkable materials.
Transient absorption (TA) spectroscopy has been used to examine electronic processes in optoelectronic materials and photocatalytic systems. Traditionally, it has been challenging to draw direct correlations between device characteristics and TA measurements. This is because the limited sensitivity of previous TA techniques required excitation intensities orders of magnitude greater than those found under operating conditions of devices. Such intense excitation can produce high densities of photo-generated charges that fill band-edge and interface states, leading to a mismatch between the electronic properties of materials examined with spectroscopy versus in devices.
Today, new noise suppression technology from Magnitude Instruments permits orders of magnitude lower excitation intensities to be used in TA measurements. For example, recent time-resolved infrared (TRIR) studies of the electronic transport properties of polycrystalline halide perovskite CH3NH3PbI3 films were reported with photogenerated charge carrier density of 3×1016 cm-3 following optical excitation at 532 nm and 500 nJ/cm2.1 This carrier density closely matched the photogenerated carrier density found in halide perovskite solar cells under one sun (AM1.5) illumination under steady-state conditions (~1016 cm-3).2
Such low excitation intensities are unprecedented in ns-TA measurements and enabled the identification of unique mid-IR electronic transitions of large polarons in the halide perovskite lattice (Figure 1A). The sensitivity of the TRIR measurements also permitted the first direct comparison of TRIR and time-resolved photoluminescence (TRPL) measurements under identical conditions (Figure 1B).
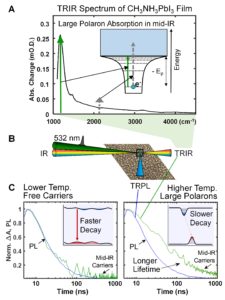
Figure 1. A. TRIR spectrum of large polarons in a halide perovskite film following band-gap excitation. The inset shows the transitions that appear in the spectrum measured at 30 ns time delay. B. Schematic showing TRPL and TRIR measurements performed under identical, device-relevant conditions. C. Overlay of TRPL and TRIR measurements reveal identical kinetics at lower temperature, indicating the presence of free carriers. Such a comparison was not possible at meaningful excitation conditions before development of new noise suppression technology. At higher temperature, dynamic disorder in the perovskite lattice localizes charges into polarons and extends their lifetime by an order of magnitude. Adapted from Ref. 1.
At reduced temperature (150 K), the data revealed that thermal motion of the perovskite lattice was sufficiently low to allow charge carriers to reside in free carrier states at the band edges of the perovskite. The TRPL dynamics exhibited band-edge emission in quantitative agreement with the dynamics of charges in free carrier states probed using TRIR (Figure 1C, left). However, at room temperature (290 K), the charge recombination lifetime increased 20-fold relative to the lower temperature (Figure 1C, right). The com-parison of TRIR and TRPL measurements under device-relevant conditions revealed that dynamic disorder due to thermal motion of the perovskite lattice led to large polaron formation. This in turn created energetic barriers to charge recombination that ultimately underpin the remarkable properties of halide perovskite optoelectronic materials.
REFERENCES
- Munson et al. Chem (2018) DOI: 10.1016/j.chempr.2018.09.001.
- Kiermasch et al. Sci. Rep. (2016) DOI: 10.1038/srep39333.

SHARE